Astrocytes, long considered as "simple" support cells for neurons, play an important role in various brain functions. They help to make the blood-brain barrier ( BBB) "watertight", protecting brain tissue (and neurons) from undesirable (toxic) molecules in the blood by allowing only the ones it needs to enter. They are involved, among other things, in neurovascular coupling, i.e. the local increase in blood flow (and energy metabolism) in regions that become activated. They are also believed to play a key role in the "glymphatic" system (as yet hypothetical), which is used to remove "waste products" that form in the brain by promoting the circulation of cerebrospinal and interstitial fluid. Its dysfunction could be responsible for the development of certain neurodegenerative diseases (such as Alzheimer's disease) due to the accumulation of aggregates in the brain. These functions are partly linked to the possibility for astrocytes to regulate water movements between the blood and the brain thanks to the presence in their membrane of a specific water channel, the aquaporin-4 (1 of the 12 types existing in the body), which control the entry and exit of water molecules. Thus, astrocytes inflate or retract as needed. In the brain only astrocytes express aquaporins (not neurons).
Remarkably, the properties and functions of astrocytes, which are six to eight times more numerous than neurons, are of major technical importance for brain imaging. Diffusion MRI (dMRI, a technique developed by Denis Le Bihan in the 1980s) explores the micro-movements of water molecules (more or less constrained depending on the local environment). It is widely used for the diagnosis of ischemic stroke in the acute phase, the reconstitution of nerve fibre pathways (tractography, the diffusion of water in axons being anisotropic and oriented in the direction of the fibres) and for the diagnosis of cancers (breast, prostate, etc.). These successes are linked to the very high sensitivity of dMRI to changes in tissue microstructure, such as the swelling of cells, astrocytes or neurons for example. It could therefore be used to study astrocytes and test their involvement in the glymphatic system in humans, and could advantageously replace the current invasive methods that consist of injecting an MRI contrast agent (gadolinium chelate) into the brain of animals to monitor its migration. However, dMRI also makes it possible to detect and locate neuronal activation (fMRI), an alternative to functional MRI by the "BOLD" effect (fMRI BOLD), which is widely used in cognitive neuroscience. The BOLD method is based on the balance between the oxy- and deoxy- forms of hemoglobin in circulating red blood cells. Due to neurovascular coupling (to which astrocytes therefore contribute strongly), neuronal activation is accompanied by a local increase in blood flow and the oxy/deoxyhemoglobin balance of the blood in favour of the "oxy" form. The resulting small change in magnetization results in maps of brain activity. Based on this neurovascular coupling, the fMRI BOLD is in fact an indirect detector of neuronal activity which is sometimes faulty and suffers from limited spatial and temporal resolution (the vascular response is several seconds out of sync with the neuronal response, which is of course considerable). fMRI by dMRI (developed by Denis Le Bihan in the 2000s with Japanese colleagues from Kyoto University) does not suffer from these drawbacks. It is based a priori on a totally different and more direct principle: the swelling of neurons (or more precisely of parts of neurons such as dendritic spines), which Denis Le Bihan has called "neuromechanical" coupling. This mechanism is, however, still hypothetical, even though Le Bihan's teams and others around the world have published a series of papers in recent years along these lines.
In two studies published in PLoS ONE, NeuroSpin researchers blocked astrocyte activity using TGN-020, an inhibitor of aquaporin 4 channels, in a mouse model and looked at the impact of this blockade on diffusion images in both fMRI and glymphatic system.
In the first study[1], at rest, administration of TGN-020 resulted in a small increase in the diffusion coefficient of water in brain tissue and a decrease in Sindex, a more sensitive marker of tissue microstructure, especially as the regions are rich in astrocytes (cortex, hippocampus). This increase would be explained by the blockage of astrocyte swelling and the resulting increase in interstitial space. The dMRI (and its Sindex) therefore seems to be a good candidate to study the contribution of astrocytes to the glymphatic system in a totally non-invasive way, possibly in humans.
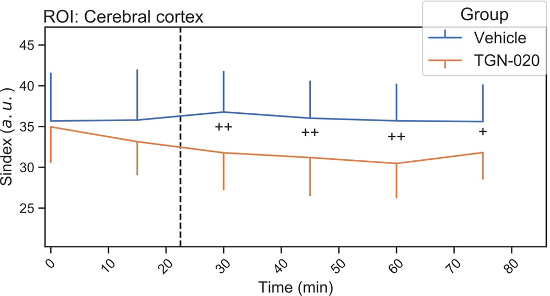
Time course of Sindex in the cerebral cortex. Blue line is vehicle group and orange line is TGN-020 group. Error bar shows standard deviation. The dashed line represents the injection time. © Debaker C. et al., PLoS ONE, 2020.
In the second study[2],during neuronal activation (visual stimulation), the authors compared the signals obtained by BOLD fMRI and diffusion fMRI. The response observed by fMRI BOLD was altered under TGN-020, confirming the role of astrocytes in neurovascular coupling at the origin of fMRI BOLD signals. It also confirms one of the limitations of this fMRI method since in fact the neuronal response remains unchanged under TGN-020, as demonstrated by electrophysiological measurements. On the contrary, the response observed by diffusion fMRI is unaltered under TGN-020, as it should be. These results confirm the independence of fdMRI from neurovascular coupling and astrocytes, and thus its direct neuronal origin. Neuronal "swelling", at the level of the thousands of dendritic spines present for each neuron, remains possible precisely because neurons, unlike astrocytes, do not have aquaporins to regulate water flow, which strongly suggests that this swelling is necessary for neuronal function.
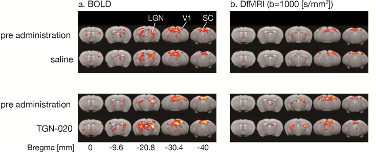
Activation maps (a : BOLD, b : IRMfD b1000) - Comparison of activation maps before and after TGN-020 (bottom pannel) or saline administration (top pannel). © Komaki Y. et al., PLoS ONE, 2020.
Contact:
Denis Le Bihan