This work was carried out within the framework of the
ISOTOPICS ITN, in collaboration with teams from INSA Toulouse (Dr.
Bruno Chaudret) and ICMMO (Dr.
Philipe Lesot), for the first article, and in collaboration with the team from INSA Toulouse (Dr.
Bruno Chaudret) and Sanofi (Dr.
Volker Derdau) for the second publication.
The selective activation of C-H bonds in complex molecules is the subject of significant research because it potentially allows these molecules to be functionalized without going through the formation of synthetic intermediates. Thus hydrogen (H), present in all organic molecules used in human health can be replaced by its stable, deuterium, or radioactive, tritium, isotope.
- In a first paper, SCBM researchers used deuterium (D) to study the modulation of the reactivity of a heterogeneous ruthenium (Ru) catalyst in carbon-hydrogen (C-H) bond activation reactions. Generally, organic ligands are used to modulate the reactivity of homogeneous, monometallic catalysts in order to increase reaction rates or to modify the regioselectivity of the chemical transformation. Here, researchers added for the first time N-heterocyclic carbenes (NHC, Figure 1A), organic ligands, to the surface of heterogeneous ruthenium catalysts to modulate their reactivity. Through a simple and robust synthesis process, they prepared in a single step new catalysts possessing various NHC grafting rates and demonstrated, by studying the nature of the products formed following incorporation of deuterium atoms, the great influence of the functionalization of the catalyst surface by NHCs on the chemoselectivity of the catalysts. Indeed, the use of modified ruthenium clusters made it possible to promote the catalysis of the reactions of CH deuteration rather than the reduction of aromatic nuclei and carbonyl compounds, reaction mainly observed when using the native Ru / C catalyst ( figure 1B). It was shown that the presence of the ligand made it possible to strongly slow down the kinetics of reduction of the aromatic rings and carbonyl functions, without modifying that of the CH activations, which made it possible to discover new reactivities for this type of Ru clusters allowing the deuteration of aldehydes.

Figure 1: Scheme of the preparation of the ruthenium catalyst modified with the organic ligand, NHC (A) and comparison of the reactivity observed with the native and the modified catalyst (B). © G Pieters/CEA
Apart from the fundamental interest of this research offering new perspectives relating to the use of heterogeneous catalysts in the context of the development of CH activation reactions, the reactions described here succeeded in labeling molecules of pharmaceutical interest with deuterium atoms (alcohols, nitrogenous heterocycles).
- The second major contribution, relating to hydrogen isotopes, concerned the development of a new method for labeling molecules of interest with tritium (T), in the context of multiple C-H activation reactions. Labeling with tritium and carbon-14 is essential for monitoring and absolute quantification
in vivo (biodistribution) or
ex vivo in animals (beta imaging and autoradiography) of a candidate drug, in order to determine its ADME properties (Absorption, Distribution, Metabolism and Excretion).
SCBM researchers describe, in the second article, a very efficient general labeling method involving an iridium pre-catalyst.This method differs from existing techniques by its ease of implementation (the commercial iridium precatalyst is stable in air and therefore easily handled), its wide field of application (extremely complex and fragile active ingredients have been radiolabeled) and the possibility of achieving record incorporation rates of tritium into the molecule. Placing under atmosphere both tritium gas and the catalyst in the presence of the substrate of interest makes it possible to generate both homogeneous complexes (figure 2A) and iridium nanoparticles capable of activating several C-H bonds on complex molecules by replacing them by C-T bonds (Figure 2B).
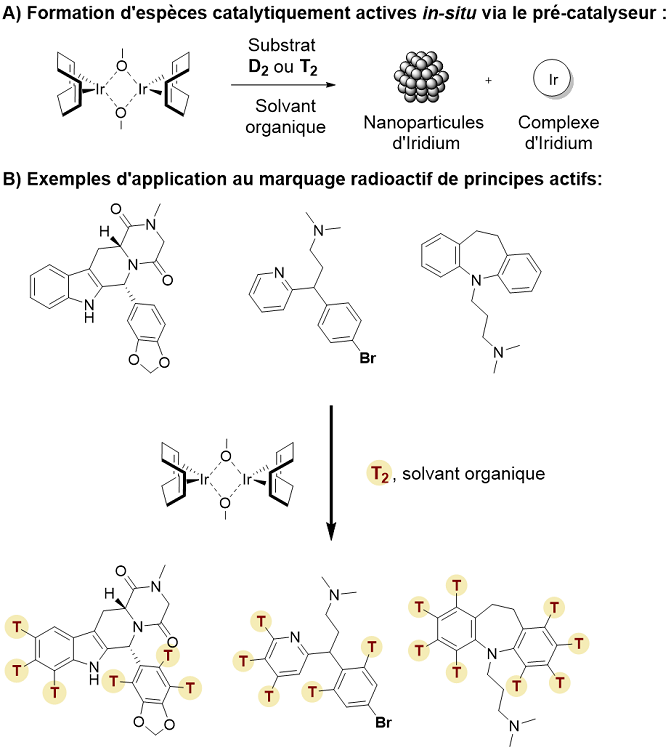
Figure 2 : Catalytic species formed in-situ in the presence of the reaction substrate and tritium gas (A) and application to the radiolabelling of active ingredients (B)
© G Pieters/CEA
No doubt these results will facilitate access to deuterated or tritiated analogues of complex molecules, essential diagnostic tools in the discovery and development of drugs.
 European funding
European funding This work by Grégory Pieters' team was carried out within the framework of the European project
ISOTOPICS, coordinated by the CEA.