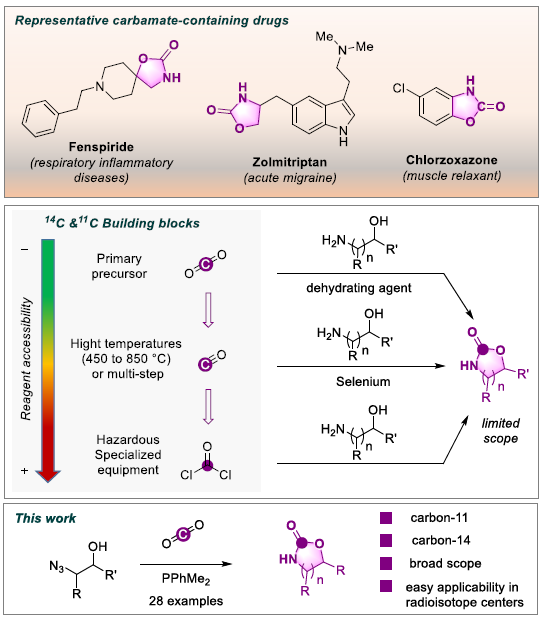
The labeling of organic molecules with radioactive carbon isotopes is essential for drug development. On the one hand, carbon-14 (14C), with a long half-life (5730 years), is used in preclinical studies to study the metabolism and biodistribution of drug candidates. On the other hand, carbon 11 (11C), with an ultra-short half-life (20 minutes), is used to label molecules used for medical imaging by positron emission tomography. These two radioactive isotopes have opposite physical properties, which generally require different strategies for their insertion into organic structures. However, given the complementary nature of their use, common labeling procedures would be beneficial for pharmaceutical R&D. In both cases, the source of carbon is the same: carbon dioxide (CO2). SCBM researchers, inspired by "click" chemistry, have already demonstrated the proof-of-concept of labeling by directly inserting radioactive CO2 (11C or 14C) into organic molecules containing cyclic ureas, in a single step and in only five minutes (see news 2018).
In a new study, published in Chemical Communications, the team shows this time that it is possible, by an equivalent method, to label with any of the carbon isotopes (11C, 13C or 14C) carbamates, urethanes that are used in the composition of pharmaceutical and phytosanitary products or plastics. In collaboration with the Service Hospitalier Frédéric Joliot (SHFJ), they have applied their method to a series of pharmaceutical molecules containing a carbamate.
In addition, a strategy of disconnection/reconnection of a functional group was implemented, thus simplifying the synthesis of the precursor and accelerating the whole process.
 European funding
European funding This work was carried out within the framework of the European project
ISOTOPICS, coordinated by the CEA.
Researcher contact: Davide Audisio (
davide.audisio@cea.fr)